Powering the Future of Clinical Research Three Clicks at a Time
TriClick™ is Hill Research's proprietary AI platform driving automation and excellence across all our CRO projects. It embodies our belief that clinical trials can be faster, safer, and smarter — powered by human expertise and intelligent systems.
Our Products
Four Core Tools, One Platform



TriClick™ Annotation
Automates CDISC-compliant annotated Case Report Forms for FDA submissions. Ensures consistency and reduces regulatory risk with AI-driven annotation at unmatched speed.
TriClick™ Compliance
Automatically desensitizes and anonymizes medical information meeting HIPAA, GDPR, and FDA requirements while preserving data utility for analysis.
TriClick™ Evidence
Extracts clinical insights from trial records, literature, and regulatory filings. Delivers real-time evidence summaries to support clinical and regulatory decisions.
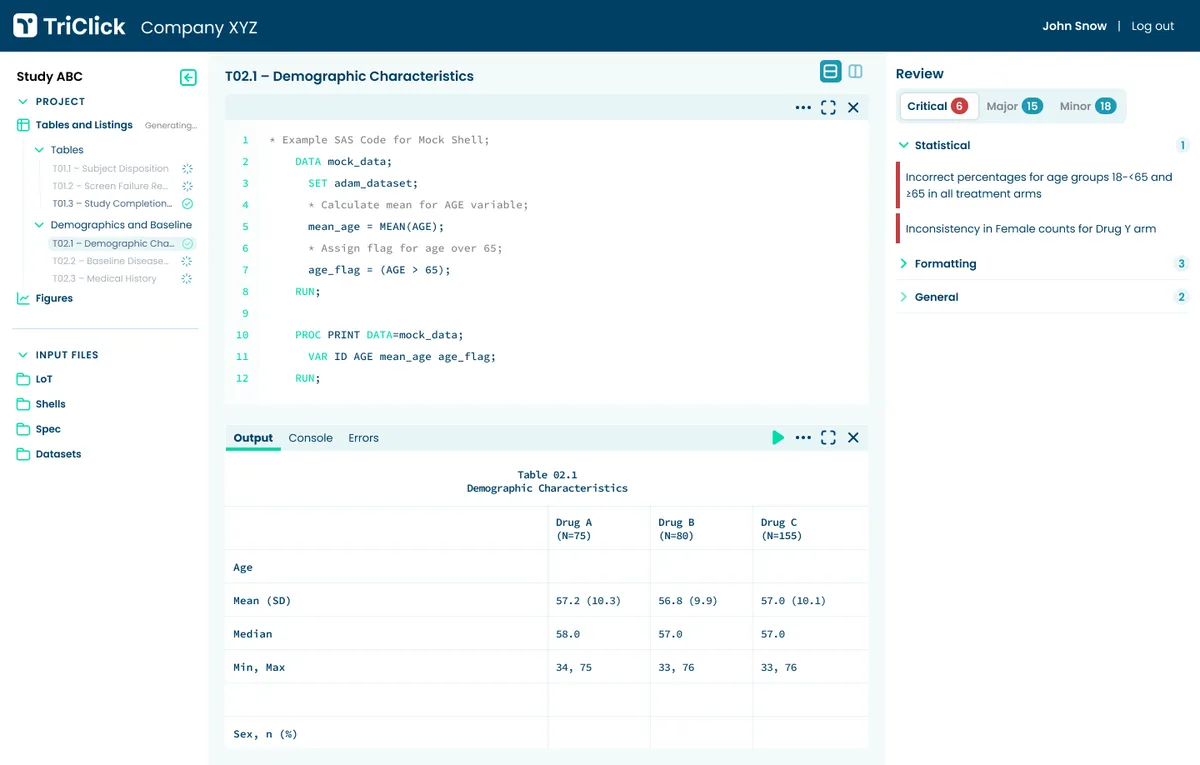
TriClick™ Stats
One-click statistical programming generating Tables, Listings, and Figures. Delivers validated outputs up to 70% faster with full SAP traceability.
AI Capabilities
6 Core AI Agents
Why Choose TriClick™
Accelerate Biometrics Workflows
100x faster workflows with 99%+ accuracy, reducing manual programming by 82%.
Ensure Regulatory Excellence
Real-time validation ensuring 100% CDISC-compliant datasets and smoother FDA submissions.
Shorten Development Timelines
Accelerating TLF delivery by 70% and enhancing coordination across functions, increasing regulatory success probability.
"TriClick™ brings the power of agentic AI into our clinical trials practice — practical, precise, and ready for today's regulatory standards. By automating critical workflows and ensuring compliance by design, it gives sponsors not just faster results, but confidence."
Frequently Asked Questions
What is TriClick™?
TriClick™ is Hill Research's proprietary AI platform that automates clinical trial biostatistics workflows. It uses specialized AI agents to execute complex biometrics tasks — including CRF annotation, compliance validation, evidence extraction, and statistical programming — in just three clicks.
How does TriClick™ ensure CDISC compliance?
TriClick™ has built-in CDISC validation at every step of the workflow. Its Compliance Agent automatically validates datasets against SDTM and ADaM standards, ensuring 100% CDISC-compliant outputs that are FDA submission-ready without manual review.
How long does it take to implement TriClick™?
TriClick™ is a cloud-based platform that integrates into existing clinical workflows without lengthy deployment cycles. Hill Research works with sponsors to configure the platform for their specific trial requirements, with onboarding typically completed within weeks.
How does TriClick™ handle data security?
TriClick™ is designed to meet HIPAA, GDPR, and 21 CFR Part 11 requirements. Its Compliance module automatically desensitizes and anonymizes medical information while preserving data utility for analysis, ensuring patient data protection at every stage.
See TriClick™ in Action
Book a consultation to discover how TriClick™ can transform your clinical trial workflows.